| Saturated Solution | ||
| Temperature єF | Temperature єC | %Salt |
| -6 | -21.11 | 23.31* |
| 0 | -17.78 | 23.83 |
| 10 | -12.22 | 24.7 |
| 20 | -6.67 | 25.53 |
| 30 | -1.1 | 26.16 |
| 32 | 0 | 26.29 |
| 32.2 | 0.1 | 26.31** |
| 40 | 4.44 | 26.33 |
| 50 | 10 | 26.36 |
| 60 | 15.56 | 26.395 |
| 70 | 21.11 | 26.45 |
| 80 | 26.67 | 26.52 |
| 100 | 37.78 | 26.68 |
| 125 | 51.67 | 26.92 |
| 150 | 65.56 | 27.21 |
| 175 | 79.44 | 27.62 |
| 200 | 93.33 | 27.91 |
| 212 | 100 | 28.12 |
| 220 | 104.44 | 28.29 |
| 227.5 | 108.7 | 28.46*** |
* Eutectic point
** Transition
point
*** Boiling point at one atmosphere pressure
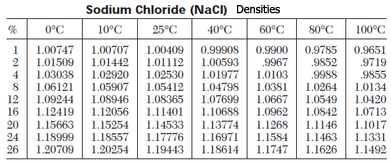
Maximum solubility of NaCl in water at 25°C is 357 mg/ml (26.3%). NaCl is unusual in that its solubility does not increase appreciably with temperature, since at 100°C, the solubility is 384 mg/ml. The density of a saturated solution at 25°C is 1.202 g/ml. A saturated solution (23% w/v) freezes at -20.5°C (5°F).
HIT THE BACK BUTTON ON YOUR BROWSER